AWS vs Azure: What’s the Difference in 2024?
Tamr: 7 Steps for Boosting R&D Outcomes with Historical Clinical Trial Data

It can cost up to $2.6 billion and take 14+ years for pharmaceutical companies to move a drug from “bench to bedside.” This is too expensive.
No longer a “one and done” effort—pharmaceutical companies are now reusing their historical clinical trials data to help inform and accelerate current clinical trials, improve the research and development process, and find new insights in old data.
How can pharmaceutical companies make the most of their historical clinical trials data?
Tamr and Mark Ramsey, former Chief Data and Analytics Officer at GSK, put together a guide on '7 Steps for Boosting R&D Outcomes with Historical Clinical Trial Data.' Download to find out strategies for:
- Aligning clinical trials data projects to key business outcomes
- Building and empowering the right team to make it work
- Driving strategies for making data an asset and priority across the organization
- Leveraging machine learning to tackle large data volume and variety in clinical trials
Recommended Content
Trending Content
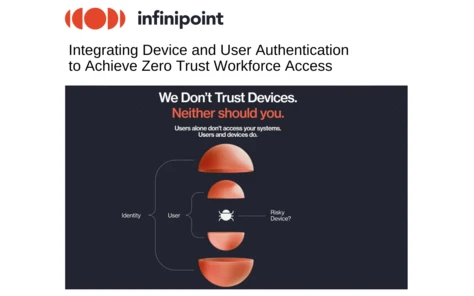
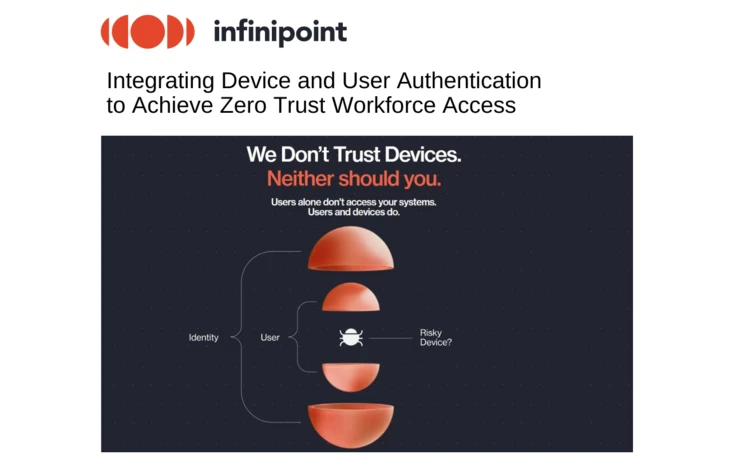
Infinipoint: Integrating Device and User Authentication to Achieve Zero Trust Workforce Access

What is Multi-Cloud Architecture? Definition & Use Cases

Infinipoint - Rethinking Workforce Access: Augmenting Authentication with Zero Device Trust

Meet MAI-1: The New AI Model by Microsoft that Rivals GPT-4

Ministry of Defence Data Exposed in China-Linked Cyber Attack